Melan Bio new veterinary drug research and development work bore fruit again
Release time:
2024-08-05
Author:
Source:
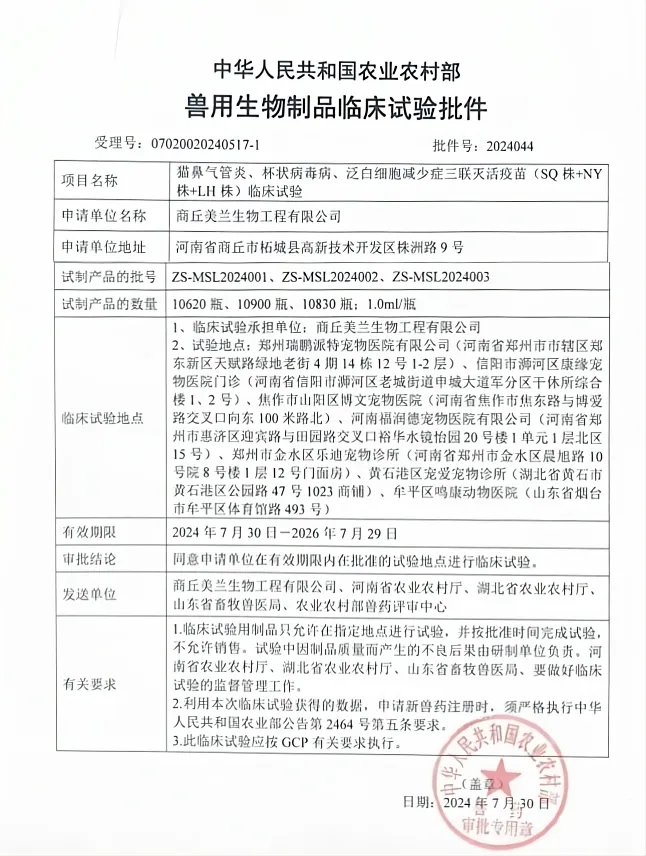
Melan Group has added another fruit of research and development. Recently, “Feline Rhinotracheitis, Cupripovirus Disease, Panleukopenia Triple Inactivated Vaccine (SQ Strain+NY Strain+LH Strain)” declared by Shangqiu Melan Bioengineering Co., Ltd. has obtained the clinical approval document from the Ministry of Agriculture, and the approval of the vaccine's clinical trial is a powerful manifestation of the company's strength in scientific and technological R&D. At the same time, this vaccine also symbolizes that the layout of the pet section of Melan Group has begun to bear fruit. The approval of the clinical trial of this vaccine is a strong manifestation of the company's strength in scientific and technological R&D, and also marks the initial achievement of the layout of the pet section of Melan Group.
RESEARCH BACKGROUND

The three strains of feline rhinotracheitis virus, feline cupripoxvirus and feline panleukopenia virus are harmful to cats and prone to mixed infections, and are the core vaccines that cats need to be immunized against. Their high contagiousness seriously affects the physical and mental health of cats, and also brings mental and economic losses to breeders. At present, although there is a cat DPT vaccine with a temporary approval number approved for emergency evaluation, it is still far from meeting the requirements of the large domestic market. In addition, vaccine immunization failures have occurred from time to time, and virus mutation is considered to be the main cause of the current vaccine immunization failure. Therefore, the preparation of a novel, high-yield cat triple vaccine against the prevalent strains has a wide range of applications for the prevention and control of cat infectious diseases and to address the domestic demand.
RESEARCH CONTENT AND ACHIEVEMENTS

In order to more effectively prevent and control feline rhinotracheitis, feline cuprovirus disease and feline panleukopenia, the vaccine research and development team of Melan Group started to conduct relevant research on the feline triple inactivated vaccine in 2018, and carried out the isolation, identification and screening of the feline rhinotracheitis virus, feline cuprovirus and feline panleukopenia virus; and optimized the cultivation of strains by adopting advanced cell suspension cultivation technology, and After spending 6 years, tens of millions of dollars and countless efforts, the triple inactivated vaccine developed can prevent three kinds of diseases in one shot, which reduces the number of vaccinations and has good feedback on the application effect, and lays the cornerstone for the development of Melan Group in the pet vaccine section.
Conclusion:
In the future, Melan Group will continue to focus on the market, customer demand for research and innovation, to provide more and more effective series of products and professional and technical services, and constantly push forward the innovation to produce more and better products and services. And actively play the advantages of advanced echelon enterprises in the industry, show corporate social responsibility, multi-dimensional and multi-faceted for the industry to fully learn to understand the correct application of biological vaccines in the industry to advanced technology advantages, improve the level of scientific and technological innovation in the industry, and work together to promote the sustainable development of the industry health, and make greater contributions to the healthy development of China's animal husbandry industry, guarding animal health and human health!
Related Blog


